What are New Approach Methodologies (NAMs) and Why are They Important?
New Approach Methodologies (NAMs) are a broad group of innovative scientific techniques designed to assess the safety and efficacy of drugs, chemicals, and other substances without relying on traditional animal testing. Since the passage of the Federal Food, Drug, and Cosmetic Act in 19381, animal testing data have been mandatory for preclinical drug evaluation. While testing drugs in animal models offers superior insights into systemic, multi-organ interactions and whole-body metabolic changes, animal models fail to fully replicate complex human physiology. Thus, the high failure rates for drugs developed in animal models are due in part to poor human predictability. Recent focus on NAMs is a paradigm shift for biopharmaceutical drug development that is moving drug safety evaluation strategies from traditional animal models to incentivizing the use of innovative human-relevant models. In 2023, the FDA Modernization Act 2.0 amended federal law to remove the mandate that all drugs undergo animal testing before human trials, allowing the use of validated NAMs for drug development. NAMs—such as patient-derived organoids, organs-on-chips, and in silico computer modeling (AI/ML)—enhance human relevance and improve safety predictions. Because NAMs do not rely on animal testing, researchers can identify toxicity earlier, reduce development costs, and accelerate timelines by providing more accurate human-specific data before expensive clinical trials.
Adopting NAMs allows researchers to move past the biological differences between species, providing human-relevant data that more accurately predicts how new drugs will work. The shift from animal testing to NAMs not only streamlines the development process but also bypasses the logistical hurdles and public sensitivities related to animal testing by using more precise, technology-driven alternatives.
New FDA Guidance on New Approach Methodologies
The FDA formally began accelerating its initiative for NAMs in a series of steps starting around 2019, with major legislative and strategic milestones occurring in 2022 and 2025, and has now published draft guidance on alternatives to animal testing. The initiative is intended to help expedite the drug development paradigm shift from whole animal studies to more relevant, safer, and more effective NAMs. In the supporting news release, the FDA Commissioner, Marty Makary, M.D., M.P.H., was quoted: “Technological advances are allowing us to move beyond animal testing in drug development, which has a poor track record of predicting safety and efficacy in humans.”
For NAMs to realize their full promise in drug discovery and personalized medicine, key characterization platforms focused on high-throughput, multivariate, and live-cell analysis are needed. Essential tools (including single-cell RNA sequencing (RNA-seq), high-content imaging and AI, functional biosensors, and microfluidics (organ-on-a-chip)) are necessary to improve reproducibility of preclinical findings across independent researchers, enable robust quality control, and validate that NAMs accurately mimic human physiology before seeking regulatory approval in drug development2.
Flow Imaging Microscopy (FIM) Is a Unique Platform for Characterizing NAMs
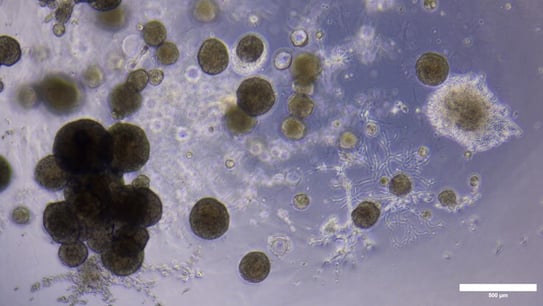
 Designed to assess particles in liquid suspension, high-throughput morphological analysis of NAMs such as 3D cell clusters, organoids, and tumoroids with FIM adds significant value to workflows by maximizing data output from limited samples and improving biological insight.
Designed to assess particles in liquid suspension, high-throughput morphological analysis of NAMs such as 3D cell clusters, organoids, and tumoroids with FIM adds significant value to workflows by maximizing data output from limited samples and improving biological insight.
Pictured here: Low-passage tumoroids imaged with brightfield microscopy (top) and with FlowCam, an FIM instrument (bottom). 3D cell clusters cultured in Matrigel® were collected and released from the scaffold matrix before imaging at 4X magnification. The observation of large 3D cell clusters and cluster aggregates with some association of extracellular matrix components is consistent between brightfield microscopy and FIM with FlowCam.
Many 3D cell culture systems, however, are not grown in suspension but are instead grown using scaffold-based systems that can make it difficult to visualize microscopic changes in 3D cell cluster behavior. While there are risks involved with removing the supporting 3D microenvironment for image analysis, monitoring morphological properties is important to ensure culture treatment conditions are appropriate for preserving clinically relevant phenotypes. Recent proof-of-concept work showed FlowCam is effective at characterizing and monitoring low-passage tumoroid models upon release from extracellular scaffolding. Measurements of morphology and cellular behavior tracked in real-time with FlowCam were consistent with data derived from manual brightfield microscopy images, demonstrating FIM as a high-throughput, non-destructive solution for quantitative characterization of precious and complex three-dimensional structures. As an image-based system, FIM provides high-resolution images, particle size distributions, and comparative analysis of 3D aggregate morphologies in real-time. FIM with FlowCam enables rapid processing time and overcomes the labor-intensive challenges of manual microscopy.
 Learn more about how FlowCam can support analysis of NAMs in our application note, “Quality Control of Organoid and Tumoroid 3D Cell Clusters with FlowCam” and contact us to initiate a conversation about your cell characterization application.
Learn more about how FlowCam can support analysis of NAMs in our application note, “Quality Control of Organoid and Tumoroid 3D Cell Clusters with FlowCam” and contact us to initiate a conversation about your cell characterization application.
References
1. Wax PM. Elixirs, diluents, and the passage of the 1938 Federal Food, Drug and Cosmetic Act. Ann Intern Med. 1995 Mar 15;122(6):456-61
2. Song X, et al. Organoids in drug development: from predictive models to regulatory integration. Drug Discovery Today. 2026; 31(2)










