Overview
Accelerate subvisible particle analysis for USP <787>/<788> workflows with FlowCam LO
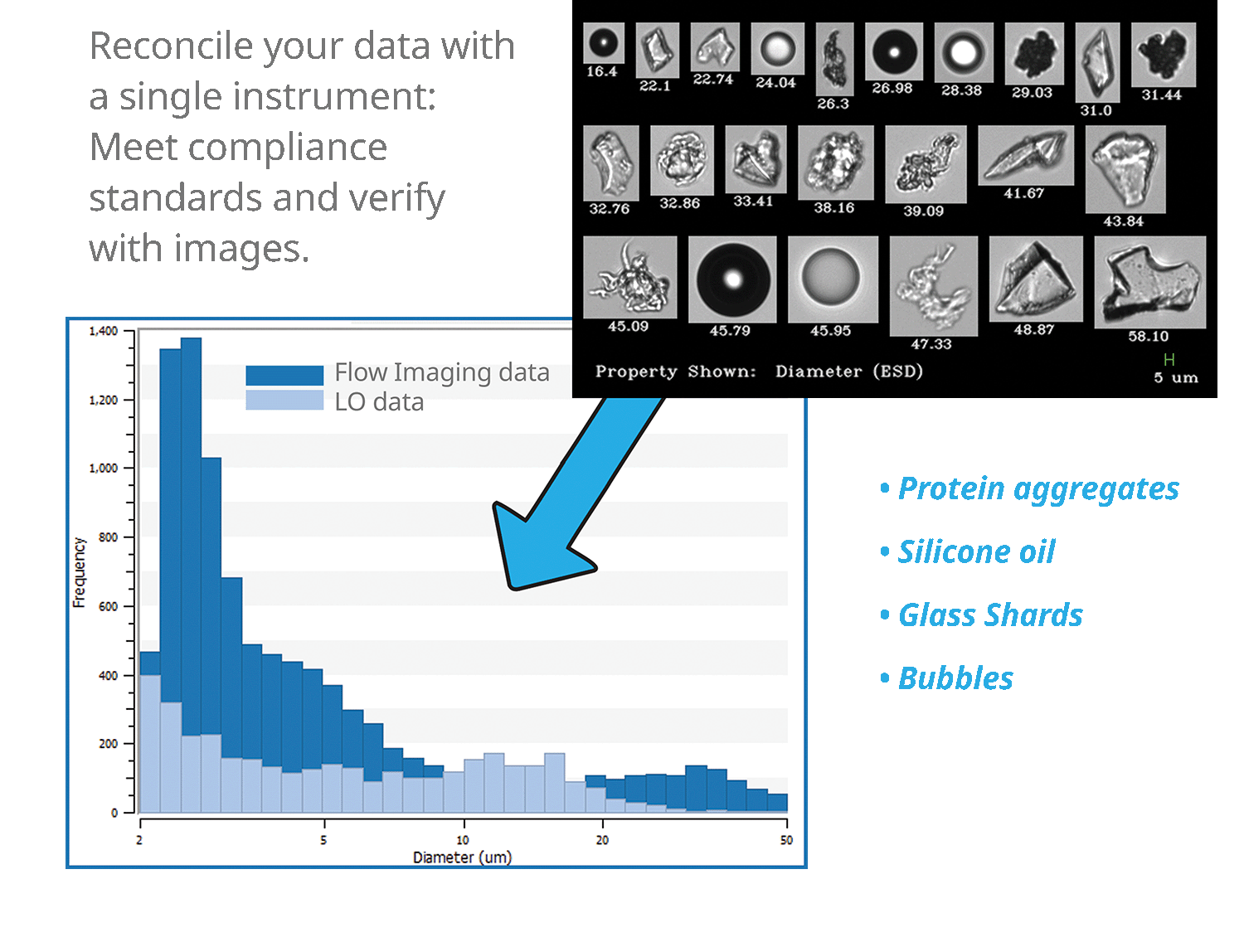
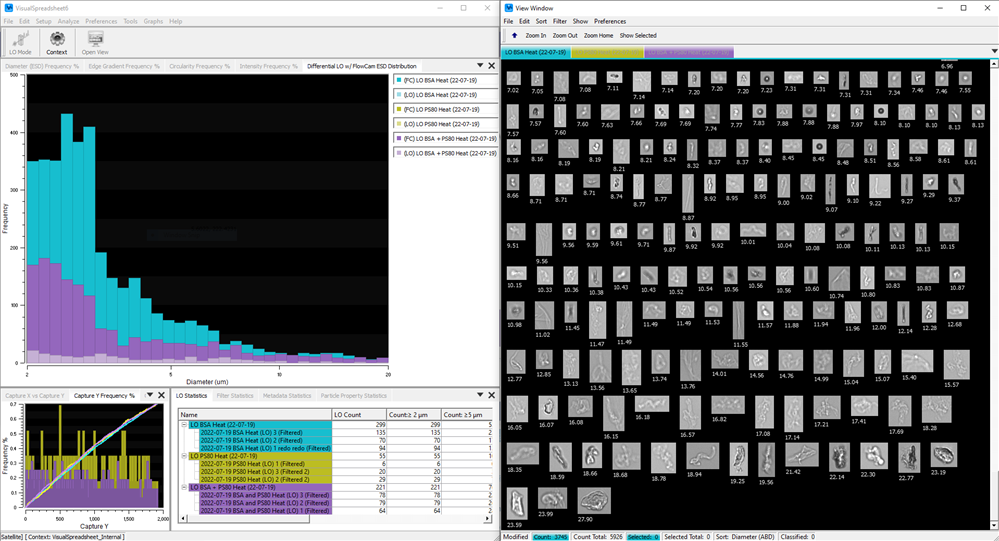
Obtain LO data consistent with USP <787>/<788> Method 1 and verify results with high‑quality FIM images — all in one instrument and one sample run.
USP <1788> recognizes FIM as an orthogonal method to LO for detection and morphological analysis of protein aggregates and other translucent low-contrast particles that do not block light.
Specifications
- Particle size range: 2 μm to 70 μm; Flow imaging module: 10X
- Minimum sample volume: 100 μL
- Sample flow rate: 0.2 mL/minute
- Camera type: High resolution (1920 x 1200 pixels) CMOS, monochrome
- Solvent compatibility: wide range of aqueous and organic fluids, including high-viscosity solvents and buffers
- Ease of use instrumentation & fully integrated VisualSpreadsheet software with optional VisualAI software module
- 21 CFR Part 11 Compliance Package available
FAQs About FlowCam LO:
What is light obscuration particle testing, and how does it work with FlowCam LO?
Light obscuration (LO) measures subvisible particles by the amount of light they block as they pass a sensor. FlowCam LO combines LO with flow imaging microscopy (FIM) in a single instrument, providing images and detailed morphological data to support and augment LO findings.
Why isn't LO particle count testing alone sufficient for subvisible particle analysis? How does it compare to flow imaging microscopy?
LO reports particle counts and sizes as equivalent spherical diameter (ESD), or the size a particle would have if it were a sphere. Because LO provides no images or shape metrics, it cannot distinguish particle type or origin. Since LO measures by light blockage, its response depends on refractive index contrast, so low‑contrast/translucent particles such as protein aggregates or silicone oil droplets can be under‑detected or incorrectly sized.
As an orthogonal method, FIM adds per‑particle images and image‑based morphology, enabling morphology‑driven classification (e.g., high-circularity silicone‑oil droplets, amorphous/irregular protein aggregates, elongated fibers, and symmetrical/optically dense air bubbles), and providing richer investigation context.
Which USP standards does FlowCam LO support?
FlowCam LO provides LO data using the USP <787>/<788> Method 1 measuring principle (light blockage), and FIM analysis recognized in USP <1788> for orthogonal characterization. Lot‑release suitability depends on your lab’s qualification, validation, and adherence to USP.
Many teams use FlowCam LO as a predictive development tool, especially under USP <787>, where smaller volumes or aliquots are relevant.
What particle size range can FlowCam LO analyze?
FlowCam LO can analyze particles in your sample from 2 μm to 70 μm.
How quickly can I run a sample through FlowCam LO, and what's the minimum sample volume required to run a test?
The minimum sample volume required to run a test with FlowCam LO is 100 μL. The sample flow rate is 0.2 mL/min.
Can FlowCam LO handle high-viscosity solvents?
Yes, FlowCam LO is compatible with a wide range of aqueous and organic fluids, including high-viscosity solvents and buffers.
What kinds of research does FlowCam LO support?
FlowCam LO combines LO and FIM to support particle profiling and risk assessment from early development through clinical manufacture. Method 1 testing (USP <788>/<787>) is commonly included in clinical lot‑release from Phase 1, while FIM provides orthogonal, image‑based morphology for screening formulation excipients and container‑closure systems, inherent/intrinsic/extrinsic differentiation, trending and investigations, custom size‑bin monitoring, process characterization, and comparability.
For biologics with limited sample quantities available for testing, these insights are especially valuable.
What training is required to operate FlowCam LO?
FlowCam LO is designed to be easy to use. We also offer a variety of training and support opportunities for new users, both during initial installation and as part of ongoing support, including:
How can I determine whether FlowCam LO is right for my subvisible particle analysis needs?
Consider FlowCam LO if any of the following apply in your case:
-
When LO alone doesn't suffice: if your subvisible particle analysis toolkit requires light obscuration (LO) readouts, and you also want images for root‑cause analysis (RCA).
-
Sample & size range: FlowCam LO is designed for typical parenteral/biologic formulations in the subvisible particles size range covered by LO, and adds particle morphology and classification in the same workflow.
-
Data integrity: the VisualSpreadsheet 6.1 Compliance Package offers GxP/21 CFR Part 11–friendly workflows and validation support for regulated labs.
-
Throughput & volume: suited for method development, stability, and lot release with small sample volumes and efficient runs.